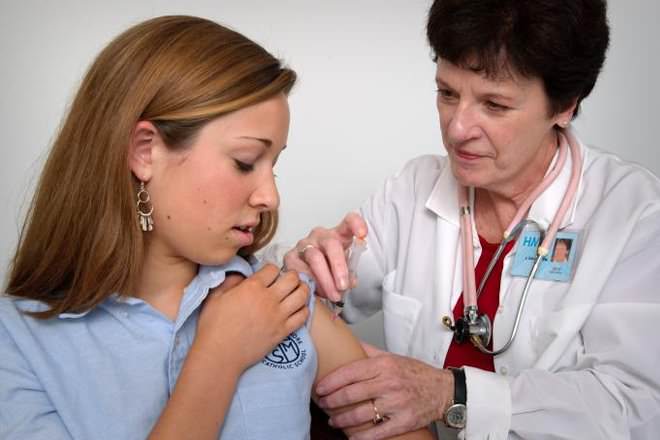
The DTaP vaccine is for children (usually infants) under 7 to inoculate them against diphtheria, tetanus and pertussis (whooping cough). Tdap is the booster shot given at age 11 and then throughout life after roughly every 10 years to ensure continued immunity through adolescence and adulthood.
Comparison chart
Form
DTaP contains an inactivated form of the toxins produced by the bacteria that causes these diseases. Tdap also contains these inactivated forms, but it contains less of the diphtheria and whooping cough toxins than the DTaP.
Vaccination Dosage
Children should receive 5 doses of DTaP: one at 2 months, one at 4 months, one at 6 months, one between 15 and 18 months, and one between 4 and 6 years.
As immunity fades over time, adults are advised to get a booster every 10 years. If they received Tdap as a child, they are advised to get the Td vaccine every 10 years instead.
Exceptions
The CDC suggest that children who are moderately or severely ill at the time they are scheduled for the vaccine should not receive it until they recover. If a child has a life-threatening allergic reaction or suffers a brain disease within seven days of receiving the vaccine, they should not have another dose.
Efficacy
A study published in the journal Pediatrics in May 2015 found that Tdap protection wanes within 2 to 4 years, a factor to which the relatively widespread pertussis outbreak in Washington state in 2012 could be attributed.[1] Many of the kids who fell sick during that outbreak were fully vaccinated per the CDC recommended schedule.[2]
Risks
DTaP carries a small risk of allergic reaction. These are seen in less than one out of a million doses, and will happen within a few minutes or hours of taking the vaccine. Symptoms include difficulty breathing, hoarseness, wheezing, hives, planes, weakness, rapid heartbeat and dizziness. Very rare side effects include seizures (example), coma and brain damage, but these are so rare that the CDC cannot tell whether they are related to the vaccine. Mild side effects include fever, redness, soreness, tiredness and vomiting.
Tdap has similar side effects.
The Vaccine Information Statement for DTap published by the CDC outlines some of the risks:
Mild problems (common)These problems occur more often after the 4th and 5th doses of the DTaP series than after earlier doses. Sometimes the 4th or 5th dose of DTaP vaccine is followed by swelling of the entire arm or leg in which the shot was given, lasting 1–7 days (up to about 1 child in 30). Other mild problems, which generally occur 1–3 days after the shot, include:
- Fever (up to about 1 child in 4)
- Redness or swelling where the shot was given (up to about 1 child in 4)
- Soreness or tenderness where the shot was given (up to about 1 child in 4)
Moderate problems (uncommon)
- Fussiness (up to about 1 child in 3)
- Tiredness or poor appetite (up to about 1 child in 10)
- Vomiting (up to about 1 child in 50)
Severe problems (very rare)
- Seizure (jerking or staring) (about 1 child out of 14,000)
- Non-stop crying, for 3 hours or more (up to about 1 child out of 1,000)
- High fever, over 105°F (about 1 child out of 16,000)
- Serious allergic reaction (less than 1 out of a million doses)
- Several other severe problems have been reported after DTaP vaccine. These include:
- Long-term seizures, coma, or lowered consciousness
- Permanent brain damage.
The VIS (Vaccine Information Statement) for Tdap published by the CDC can be found here.

 Antibiotics
Antibiotics  Amoxicillin
Amoxicillin  Bacteria
Bacteria  Minerals
Minerals  Vitamin B
Vitamin B  Vitamin D2
Vitamin D2
Comments: DTaP vs Tdap