Butane and methane are hydrocarbons from the same chemical family compounds known as alkanes. They are components of natural gas and extraction of oil.
Comparison chart
| | Butane | Methane |
|---|---|---|
| CAS number | 106-97-8 Y | 74-82-8 Y |
| PubChem | 7843 | 297 |
| ChemSpider | 7555 | 291 |
| SMILES | CCCC | C |
| InChI | 1/C4H10/c1-3-4-2/h3-4H2,1-2H3 | 1/CH4/h1H4 |
| Molecular formula | C4H10 | CH4 |
| Molar mass | 58.12 g mol−1 | 16.042 g/mol |
| Appearance | Colorless gas | Colorless gas |
| Density | 2.48 kg/m3, gas (15 °C, 1 atm) 600 kg/m3, liquid (0 °C, 1 atm) | 0.717 kg/m3, gas 415 kg/m3 liquid |
| Melting point | −138.4 °C (135.4 K) | -182.5 °C, 91 K, -297 °F |
| Boiling point | −0.5 °C (272.6 K) | -161.6 °C, 112 K, -259 °F |
| Solubility in water | 6.1 mg/100 ml (20 °C) | 35 mg/L (17 °C) |
| MSDS | External MSDS | External MSDS |
| NFPA 704 | 4 1 0 | 4 1 0 |
| Flash point | −60 °C | -188 °C |
| Explosive limits | 1.8 - 8.4% | 5 – 15% |
| Related alkanes | Propane; Pentane | Ethane, propane |
| Related compounds | Isobutane; Cyclobutane | Methanol, chloromethane, formic acid, formaldehyde, silane |
| Structure and properties | n, εr, etc. | n, εr, etc. |
| Thermodynamic data | Phase behaviour Solid, liquid, gas | Phase behaviour Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS | UV, IR, NMR, MS |
| Uses | Butane is used in refrigeration, cigarette lighters, heating as LPG or liquefied petroleum gas. | Methane is an energy source used for heating purposes. |
| Harmful effects | Butane can cause asphyxiation and ventricular fibrillation. | Methane is a potent greenhouse gas. It can form explosive mixtures with air. |
| Sources | Crude oil, natural gas. | Natural gas fields, biogas production, atmospheric methane, extra terrestrial methane. |
Chemical formula and Molecular structure of methane vs butane
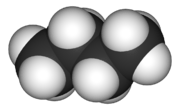
Butane is C4H10 while methane has the chemical formula – CH4. Thus there are four hydrogen atoms in a methane molecule while there ten hydrogen atoms in a butane molecule. The molecule of methane forms a tetrahedral structure while butane is a linear structure.
Chemical Reactions of methane and butane
Butane reacts with oxygen to form carbon dioxide and water vapor. Under conditions of limited oxygen, butane can form carbon monoxide or charred carbon. It reacts with chlorine to give chlorobutanes and other derivatives. Methane undergoes combustion to give formaldehyde, carbon monoxide and finally carbon dioxide and water vapor. The process is known as pyrolysis.
Ease in Handling
Both methane and butane are odorless gases at room temperatures. Butane can be easily liquefied hence it is sold as fuel for camping and cooking. It is mixed with propane and other hydrocarbons to from LPG which is commercially used for heating and cooking purposes. Methane is difficult to transport and is transported by pipelines and LNG carriers.
Unlike methane which is a stubborn gas in normal pressure and temperature, butane turns into a liquid when compressed. This property is attributed to the weak central carbon atom bonds. As soon as this liquefied gas comes in contact with air, it reacts with ignition source and become inflammable.
Isomers
Butane exhibits structural isomerism unlike methane and has two isomers, n-butane and iso-butane. Methane does not exhibit isomerism.
Uses of methane vs butane
Butane is used in deodorants, cigarette lighters, cooking and heating gas cylinders, propellant in aerosol sprays and refrigerants etc. Methane also known as marsh or swamp gas is used for firing electrical generation stations, power automobiles etc.
Sources
Methane is found in wetlands and oceans, atmosphere, human sources like burning fuel, raising livestock, fermentation of organic matter etc. Butane can be obtained as a byproduct during extraction of crude oil and is a component of natural gas.



 Oxygen
Oxygen  Ionic
Ionic  CNG
CNG
Comments: Butane vs Methane