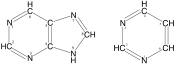
A nucleoside consists of a nitrogenous base covalently attached to a sugar (ribose or deoxyribose) but without the phosphate group. A nucleotide consists of a nitrogenous base, a sugar (ribose or deoxyribose) and one to three phosphate groups.
Nucleoside = Sugar + Base
Nucleotide = Sugar + Base + Phosphate
Comparison chart
| | Nucleoside | Nucleotide |
|---|---|---|
| Chemical Composition | Sugar + Base. A nucleoside consists of a nitrogenous base covalently attached to a sugar (ribose or deoxyribose) but without the phosphate group. When phosphate group of nucleotide is removed by hydrolysis, the structure remaining is nucleoside. | Sugar + Base + Phosphate. A nucleotide consists of a nitrogenous base, a sugar (ribose or deoxyribose) and one to three phosphate groups. |
| Relevance in medicine | Several nucleoside analogues are used as antiviral or anticancer agents. | Malfunctioning nucleotides are one of the main causes of all cancers known of today. |
| Examples | Examples of nucleosides include cytidine, uridine, adenosine, guanosine, thymidine and inosine. | Nucleotides follow the same names as nucleosides, but with the indication of phosphate groups. For example, 5'-uridine monophosphate. |
Biological Function
Nucleotides are building blocks of nucleic acids (DNA and RNA). A nucleic acid contains a chain of nucleotides linked together with covalent bonds to form a sugar-phosphate backbone with protruding nitrogenous bases. For example, DNA contains two such chains spiraling round each other in the famous double helix shape. The two chains in the double helix are held together along their length by hydrogen bonds that form between the bases on one chain and the bases on the other.
The biological functions of nucleotides are:
- Data storage - as part of DNA/RNA
- Energy Currency - ATP
- Cellular communication (cAMP; ATP allosteric regulator)
- Co-enzyme catalysis
This video explains the difference between nucleotides, nucleosides and nucleic acids.
Relationship
When nucleosides are phosphorylated by specific kinases (a type of enzyme in the cell on the sugar's primary alcohol group (-CH2-OH), nucleotides are produced.
Nucleotidases are hydrolytic enzymes which break down nucleotides (such as the thymine nucleotide) into nucleosides (such as thymidine) and phosphate.

 Purines
Purines  DNA
DNA  Replication
Replication  Genotype
Genotype  Allele
Allele  Frederick Griffith
Frederick Griffith
Comments: Nucleoside vs Nucleotide